Prevalence, Past Experience, and Competition
Prevalence
HIV (Human Immunodeficiency Virus) attacks the body’s immune system and if left untreated, it can lead to AIDS. Currently, there is no cure but the advanced treatments available nowadays can make it possible for patients to live a long life and not transmit the virus to their partners.
The majority of HIV patients live in low- and middle-income countries. In 2019, there were 20.7 million people with HIV in Eastern and Southern Africa, 4.9 million in Western and Central Africa, 5.8 million in Asia and the Pacific, and 2.2 million in Western and Central Europe and North America according to HIV.gov.
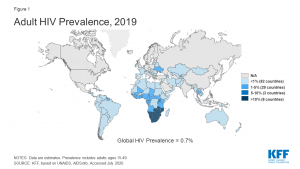
Map by KFF.org.
Countries with Past Experience in HIV Clinical Research
The way we usually measure the research experience of a given country is by calculating the number of trials performed per one million people. The map below shows, on a global level, which countries have had more clinical trials than others based on their population:
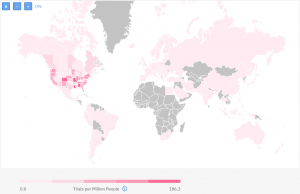
The darkest red regions are the ones with the most previous experience in running studies in HIV. Historically, this has been the USA.
However, when we exclude Phase 4 and, if we only want to see trials conducted in the past 3 years, the map becomes more relevant to the current clinical research landscape:
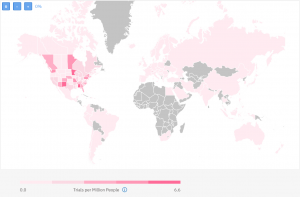
British Columbia and Manitoba have emerged as experienced destinations for clinical research in HIV.
However, if we look at the number of trials completed in general divided by the number of people who live there, then Puerto Rico is leading the rankings, followed by some European countries:
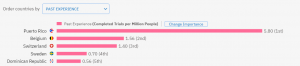
Interestingly, even if we remove the time limitations, Puerto Rico still ranks first. Do you want to know which are the next 4 countries? You can check on TrialHub.
Interesting Note:
Among the 7,488 analyzed trials, we identified 193 trials that had a virtual component included in their design. 40 of those research studies are Treatment focused. You can find their details on TrialHub.
Countries with Active or Recruiting Clinical Trials
At the moment there are 1,790 active, recruiting, or not yet recruiting clinical trials. Of them, the ones currently recruiting patients are 876.
The countries with the highest number of trials are the USA, Spain, Italy, and the UK. However, if we want to better measure the competition level when it comes to patient recruitment, it’s best to measure trials versus population. Based on trials per million people, the top 5 countries with the highest competition are as follows:
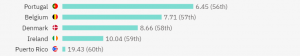
On the below map, you can also get a better idea of the global competition in running HIV clinical trials:
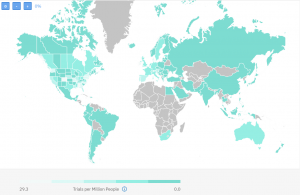
The dark green regions are the ones with less or no competitive trials.
As you can see on the map, there are regions with less competition even within the USA where historically research has been focused. Also, some prevalent regions currently have unoccupied sites which could be an opportunity for engaging their patient population.
To learn more about such opportunities – including the Patient Pathway in a given country, as well as Standard of Care – you can reach out to our team at patientsfirst@findmecure.com.
Recruitment Rates and Trials’ Performance Statistics
We have analyzed 5,566 clinical trials for their Recruitment Rates and assessed countries based on their Average Recruitment Rate (patients/ site/ month). The map below represents the regions with high ARR in dark yellow, ranging between 14.42 patients/site/month and 0.09 patients per site per month.
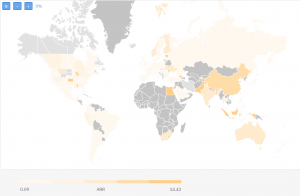
For Phase 3 studies only, ARR varies between 0.09 and 2.52 and for Phase 1 and 1/2 between 0.04 and 8.3 patients/site/month.
Looking at the highest recruiting countries, these are the top 5:
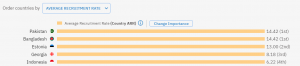
On the other end of the ranking, the countries that have the lowest ARR are:
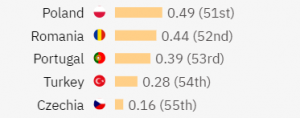
All these rates are based on all sorts of research studies. If you’d like to know how the ranking changes for treatment-focused studies, you can log into TrialHub or get in touch with us to discuss and align your data needs with your protocol: patientsfirst@findmecure.com.
Interesting Stats:
Here are some statistics on the planned vs. actual performance of HIV clinical trials:

On average, HIV trials finished with the same number of sites they started with. This could be attributed to being only 8% off in their ARR estimations. In this case, delays cannot be attributed to patient recruitment challenges.
If you’d like to know more, you can check on TrialHub.
Sites and Investigators
At TrialHub, we have identified 5,908 research centers that have been working on clinical trials for HIV. You can see on the map how they are distributed. 4,144 of them have had at least one study focused on treatment.
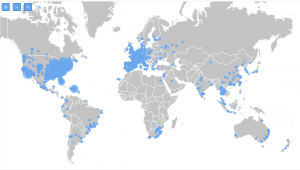
Highlighted in red are the ones currently working on trials in HIV:
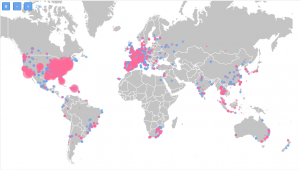
When it comes to research sites that are currently not involved with studies in this indication, out of 5,908, we identified that 4,115 are currently not occupied.
Below, we provide you with a list of the most experienced organizations in HIV research in the countries with the highest number of trials per million people:
- Univ of Puerto Rico / Univ Children’s Hosp AIDS, Puerto Rico
- Centre Hospitalier Universitaire St. Pierre (C.H.U. St. Pierre), Belgium
- University Hospital Zurich, Switzerland
There also are many investigators with backgrounds in HIV research. At TrialHub, we identified 7,756 such investigators. 3,852 of them have experience in trials focused on researching treatments. 553 of these investigators have worked on Phase 1 and Phase 1/2 studies.
Interesting Note:
The top 10 investigators with the most completed clinical trials in HIV are:
- Jaime Martinez – United States, Illinois, Chicago
- Patricia Emmanuel – United States, Florida, Tampa
- Donna Futterman – United States, New York, Bronx
- Marta Boffito – United Kingdom, London
- Lawrence Friedman – United States, Florida, Miami
- Barbara Moscicki – United States, California, San Francisco
- Ellen Abdalian – United States, Louisiana, New Orleans
- Marvin Belzer – United States, California, Los Angeles
- Bonaventura Clotet – Spain, Barcelona, Badalona
- Irma Febo – Puerto Rico, San Juan
To learn more about them, and other investigators available to run clinical trials in HIV you can contact us at patientsfirst@findmecure.com.
TrialHub is a feasibility intelligence platform that supports feasibility, startup, patient recruitment and clinical project managers with a 360-degree overview of the clinical research landscape in order to plan successful and on-time clinical trials.
TrialHub does this by aggregating thousands of data sources in real-time and combining them with local experts’ insights.
The data provided in this article is gathered in November 2020 and is based on 17 clinical trial registries (clinicaltrials.gov, eudract.ema.europa.eu etc.) and analytics about thousands of clinical trials.
If you want to get an up-to-date and/or customized feasibility for HIV or need an assessment about a different indication or TA, please contact us at patientsfirst@findmecure.com

